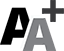Dr. Robert Montgomery, director of NYU Langone’s transplant institute, prepares a pig kidney for transplant into a brain-dead man in New York on July 14. Researchers around the country are racing to learn how to use animal organs to save human lives. AP Photo/Shelby Lum
There’s long been a gap between the relatively small number of organs available for transplant and the long waiting lists of potential recipients. Last week, the world got a little closer to a future in which pigs — yes, pigs — could narrow that gap.
A new study, published in Nature, showed that a monkey lived for two years after receiving a gene-edited pig kidney. The remarkable feat, one of several this year in the once-stagnant field of animal-to-human transplantation (also known as xenotransplantation), is an important step. We might finally be at a point where edited organs from one species can help patients of another.
Of course, so far nearly all of those patients are monkeys. The critical next phase: clinical trials in humans. That is a big leap, to be sure. But researchers are quickly amassing enough data from primates — and from limited experiments with people — to support the eventual launch of small, careful studies in humans.
The need is certainly sufficient to justify the carefully calculated risks a human trial would entail. Every day, 17 people die in the U.S. while waiting for an organ transplant. Even if everyone in the country signed their donor card, there still wouldn’t be enough viable organs to meet the need.
“There are patients out there who are going to die and not get a transplant,” says Megan Sykes, director of the Columbia Center for Translational Immunology. “For the right patient group, it might be appropriate to move ahead with some of these trials.”
Doctors have spent decades dreaming of engineering pigs to produce kidneys, hearts, livers and pancreases that could function in humans. Why pigs? Because they’re easily bred and can grow organs that are the right size for humans. And in theory, anyone who enjoyed a BLT would find it hard to raise an ethical objection to rearing a pig to save a human life. Moreover, the company behind last week’s study believes the same gene-edited pig could donate multiple organs — a kidney to one patient, a heart to another, a liver to a third.
Crispr, the Nobel Prize-winning gene-editing tool, has moved the field forward by making it easier to improve the viability of the organs. The pig kidneys featured in this week’s study were designed by eGenesis, a biotech firm cofounded by Harvard bioengineer George Church.
The monkey that survived for two years post-transplant received a kidney from a pig with a whopping 69 edits to its genome. That’s a vastly different approach from the mere handful — typically three or fewer — modifications most other xenotransplantation teams are making to organs. Some question whether dozens of edits are necessary, and if it will be hard to consistently reproduce so many changes. A high number of edits also makes it hard to parse which of the added or subtracted genes were most beneficial.
Despite such questions over eGenesis’s choices, xenograft experts seem to all agree that the company’s study helps bring clinical trials closer to reality.
Researchers have already done some human tests; several teams have transplanted engineered pig kidneys into brain-dead patients, where they’ve been able to demonstrate the organs have some function. (Typically people who had been organ donors, but whose organs turned out to be nonviable for donation, so their families chose to donate their bodies to science. And for ethical reasons, these experiments are kept short — to days or weeks.)
And the University of Maryland Medical Center has given genetically modified pig hearts to two people with terminal heart disease. The first recipient died after two months, but the hope is the second, who is just three weeks out from the procedure, will get a little more time. In an interview, his doctors told me that the patient is now able to stand and has been asking about the latest sports scores.
Those experiments have been done as one-offs, under compassionate use exemptions granted by the Food and Drug Administration that allow patients who’ve run out of options to consent to medical experiments. So what, then, will it take to get to clinical trials? Mike Curtis, eGenesis’s CEO, told me that the FDA’s consistent stance has been that, before human trials can start, the company needs to show its pig kidneys are safe and effective in a monkey for a year.
That seems reasonable. Anyone volunteering for a trial would want to know the experimental organ would give them a real chance to gain meaningful time.
For eGenesis, those trials for kidneys are still likely another two years off, but tests for its pig liver, which is intended to work just long enough to let people heal or get a human donor, could come sooner.
With every new experiment, the field learns more about how to design organs, who might benefit most, and how to prevent the recipient’s immune system from rejecting the new organ. And each is a step toward a future where genetic engineering can eliminate the gap between need and availability of organs. That future may still feel fantastical, but it’s getting closer every day.
Lisa Jarvis is a Bloomberg Opinion columnist covering biotech, health care and the pharmaceutical industry. Previously, she was executive editor of Chemical & Engineering News.
Send questions/comments to the editors.